 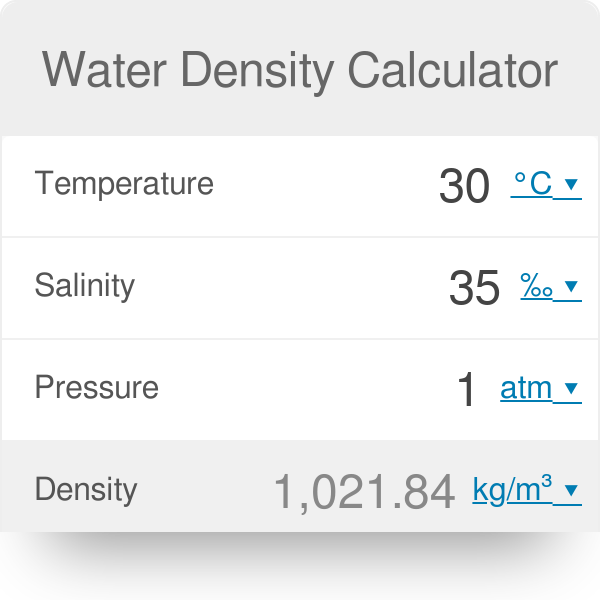
You can quickly evaluate the temperature of water while it is cooling down. You can start by heating your mixture (e.g., in a microwave), then submerging your objects in it. Check out the Advanced mode to see how much pure water and salt you need to obtain salt water with a specific density. Although changing the external pressure is difficult, you can create a liquid with known salinity and control its temperature. The same density of water is 1.0249 g/ml or 63.982 lb/ft³.īut this is not the end! Perform your own experiment at home – take a few objects with an unknown density (but more or less equal to the value for the density of water, i.e., 1000 kg/m³). And that's it! The density of salt water is 1,024.9 kg/m³.What is the density of water at 20 ☌ of salinity S = 35‰ and under the pressure of 1 atm? Let's give our water density calculator a try and find out! However, we can spot some common properties – the density of salt water increases for higher salinity and external pressure. Here ρ(T) is the density of pure water derived from the previous chapter. 
Still, generally, it uses the same concept as the equation for the temperature dependence of density – a polynomial of mixed terms of temperature, salinity, and pressure: The overall formula is complicated, so we won't show it here explicitly. In our density of water calculator, we use a method proposed by Millero and his coworkers. This quantity is usually given in per mille ‰, parts per thousand (ppt), or practical salinity units (psu), which are basically equivalent. In other words, mixing a mass of pure water, m₀, with a mass of salt, m₁, gives you the salinity S of salt water. Where m₀ is the mass of pure water and m₁ is the mass of salt. The basic parameter that tells us about the amount of salt in salt water is salinity, S, given as: The mixture of water and salt hereafter referred to as salt water has a different density than pure water. 
Although the outcome is an approximation, it's provided with sufficient precision. We use scientific notation to express all of the small values more clearly. Where the temperature T is in ☌, and the values of coefficients are the following: To get around this, our water density calculator uses an approximate equation based on the 5 th order polynomial: While there are tables of pure water density between water's freezing point ( 0 ☌ or 32 ☏) and its boiling point ( 100 ☌ or 212 ☏), there isn't a straightforward formula which yields the exact value for a given temperature. Water has an intriguing property – it reaches its maximum density at about 4 ☌ or 40 ☏. Qualitatively, it's fairly simple, but from a mathematical point of view, estimating the outcome is a different story. As a result, the mass stays constant, but the volume increases, causing the density to decrease. #V_"water" = color(green)(|bar(ul("10.You probably know of the phenomenon of the density of water changing at different temperatures, even if you've never thought about it in scientific terms ever wondered why ice floats? This change is mainly caused by thermal expansion – where the same amount of substance occupies more and more space as the temperature increase. Round to three sig figs, the number of sig figs you have for the mass of the sample, the answer will be You can use the density of water as a conversion factor to determine what volume would #"9.98 g"# of water occupy at this temperature So, at water is said to have a density of #"0.998203 g mL"^(-1)#. I assume that the problem provided you with a table of water densities a various temperatures, but since you don't have it listed here, I'll use this resource to find it This means that you will get less than #"1.00 g"# per milliliter of water as you increase temperature. So, as temperature increases, the density of water decreases. Water has a maximum density of approximately #"1.00 g mL"^(-1)# at #-># you can read more about that in this Socratic answer. This is why the problem provides you with the temperature of the water. The idea here is that water's density is temperature dependent, meaning that it varies depending on the temperature of the water sample. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
